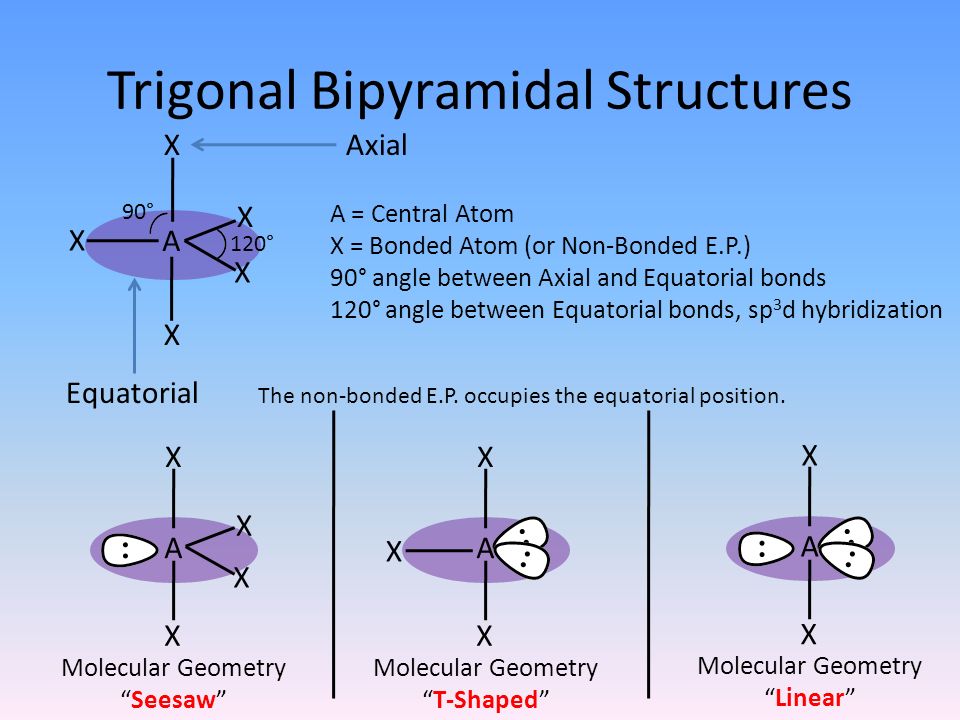
This is not consistent with experimental evidence. Figure 1. The hypothetical overlap of two of the 2 p orbitals on an oxygen atom (red) with the 1 s orbitals of two hydrogen atoms (blue) would produce a bond angle of 90°. The prediction of the valence bond theory model does not match the real-world observations of a water molecule a different model is needed. Experimental evidence shows that the bond angle is 104.5°, not 90°. If this were the case, the bond angle would be 90°, as shown in Figure 1, because p orbitals are perpendicular to each other. Valence bond theory would predict that the two O–H bonds form from the overlap of these two 2 p orbitals with the 1 s orbitals of the hydrogen atoms.
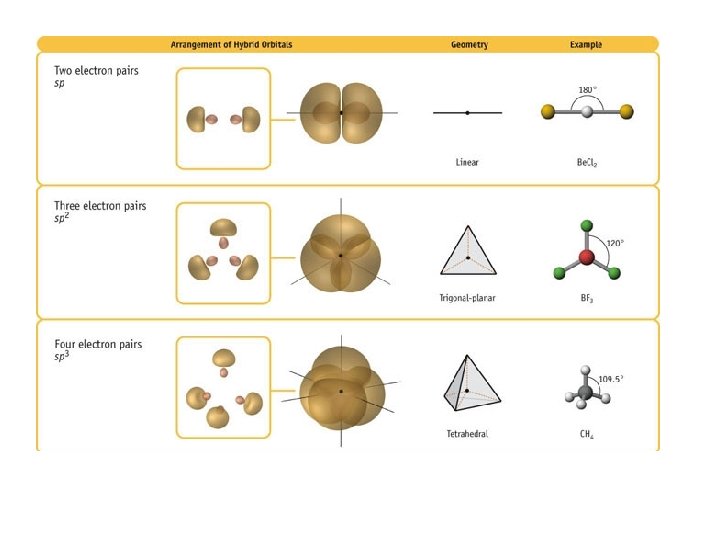
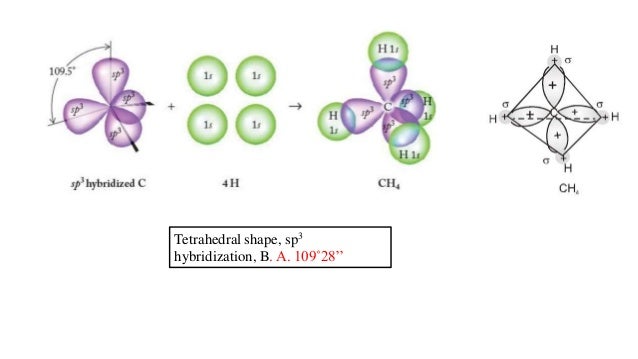
Oxygen has the electron configuration 1 s 22 s 22 p 4, with two unpaired electrons (one in each of the two 2 p orbitals). As an example, let us consider the water molecule, in which we have one oxygen atom bonding to two hydrogen atoms. However, to understand how molecules with more than two atoms form stable bonds, we require a more detailed model. Thinking in terms of overlapping atomic orbitals is one way for us to explain how chemical bonds form in diatomic molecules. Determine the hybrid orbitals associated with various molecular geometries.Explain the concept of atomic orbital hybridization.The exact information has been introduced in several other theories.By the end of this section, you will be able to: These assumptions are made using VSEPR theory. Let’s find the molecule having $$ and the shape is either trigonal bi pyramidal or octahedral geometry. So the atoms in a molecule get separated from each other to avoid the repulsion.
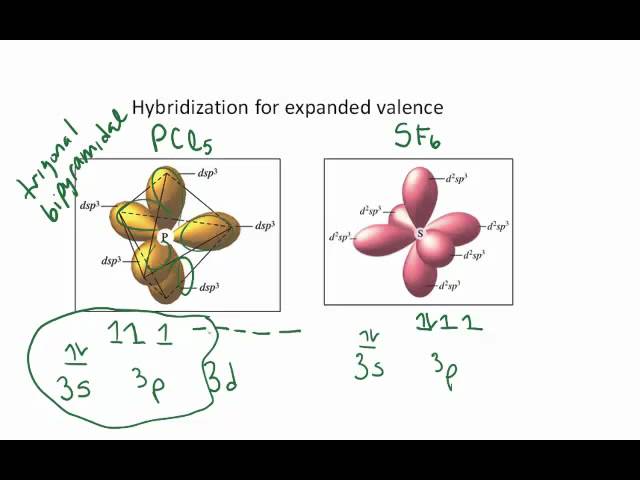
The main principle of this theory is that the electrons repel each other. valence shell electron pair repulsion theory. Hint:Hybridization of a molecule can be determined using VSEPR theory, i.e.
